|

by Dr. Joseph Mercola
February 25, 2026
from
Mercola Website
PDF Format
Story at-a-glance
-
Heart attacks occur every 40 seconds in America,
affecting a total of 805,000 people annually. It is
characterized by blocked coronary arteries that starve
cardiac muscle of blood flow
-
Australian researchers found human hearts can regenerate
muscle cells after heart attacks, with preserved cardiac
tissue showing 7% to 8% mitosis rates (a measure of cell
regeneration activity), though 25% to 50% is needed for
full repair
-
Hypoxia, which is the oxygen-deprived state during heart
attacks, may also trigger regeneration, similar to how
fetal hearts produce new cells in the low-oxygen womb
environment
-
Advanced heart failure reduces heart muscle cell renewal
dramatically, but patients with mechanical heart pumps
showed regeneration rates of 3.1% annually - six times
higher than healthy hearts
-
Prevention remains crucial. Strategies such as
minimizing linoleic acid consumption, monitoring body
fat percentage, engaging in moderate resistance
training, and learning to recognize heart attack warning
signs increase outcomes
|
According to the U.S. Centers for Disease Control and Prevention
(CDC), a heart attack occurs every 40 seconds throughout America.
This totals to around 805,000 people every year - 605,000 of them
experience it for the first time, while the remaining 200,000 are
repeat cases.
Moreover, 1 in 5 people don't know they've already had
a heart attack. 1
But what exactly happens when you have a heart attack?
Simply put,
blood flow to the heart becomes severely restricted usually due to a
buildup of plaque in the coronary arteries.
Once a complete blockage occurs, cardiac
muscles die as they also don't get blood flow.
From here, symptoms such as,
chest pain, cold sweats, fatigue, nausea, and
shortness of breath manifest. 2
Treatment is centered on restoring blood flow as soon as possible to
prevent further tissue death.
Here lies a question that has bothered
researchers for years now - once a heart attack occurs,
can cardiac
tissue regenerate on its own and achieve optimal function again?
New
evidence shows that there's a sliver of hope, but it needs to be
fleshed out further.
The Human Heart can Regrow
Cardiomyocytes after a Heart Attack
Experts have long been aware that certain animals can regrow their
own heart cells after a heart attack.
One example is zebrafish,
which can actually do a complete regrowth.
Meanwhile, mice have
shown the ability to induce mitosis (dividing and multiplying of
cells) in the affected area.
The
human heart, on the other hand, was believed to be different.
According to Sean Lal, Ph.D., a professor
of clinical and molecular cardiology at the University of Sydney and
coauthor of the featured study, medical students are generally
taught that the number of heart cells you're born with remains the
same throughout your lifespan or until you suffer a heart attack.
3
Now, 4 a team of Australian researchers found that
this may not be
the case.
Their study (Human
Hearts intrinsically Increase Cardiomyocyte Mitosis after Myocardial
Infarction), published in the journal Circulation
Research, made a big breakthrough that deepens the understanding of
what we know about the human heart.
Specifically, they discovered
how it can regenerate new heart muscle cells (cardiomyocytes).
5
To test their hypothesis, the researchers used a heart that sat in
storage for almost two decades.
It was donated by the family of a
48-year-old man who suffered from a severe heart attack. He was
brain-dead and on life support, but the damaged heart couldn't be
transplanted into someone else.
The heart was preserved and frozen in liquid nitrogen to preserve
tissue quality.
"Essentially, the tissue and cells were
'frozen in time'," according to lead researcher Rob Hume, Ph.D.
6
-
Analysis of the heart
Using an array of analytical techniques,
the researchers were able to determine how the heart
underwent mitosis.
According to Lal, the samples they
collected from the donor heart showed a mitosis rate of 7%
to 8%. But to be able to repair the heart back
to its optimal state, the mitosis rate should ideally be 25%
to 50%.
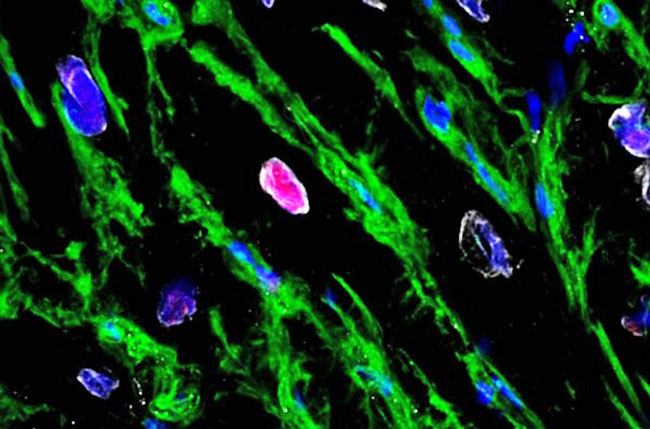
In the image below, you can see that the pink area is where
a cardiomyocyte is regenerating.
This was triggered by
adding certain antibodies into the tissue, which attached to
proteins that are expressed during mitosis:
Source:
The Age,
January 18, 2026
-
A theory on why regeneration
happens
Lal explained that hypoxia could be the
factor that triggers mitosis in the heart muscles.
Basically, the very same oxygen-deprived environment caused
by a heart attack also triggers regeneration in the affected
area.
This supports his initial theories
regarding fetal hearts, noting that,
"Fetal hearts make tonnes of new
heart cells in utero, which is an oxygen-low
environment."
He connects this to his research about
adult hearts: 7
"It's almost like the heart has some
inbuilt memory.
Maybe when you have low oxygen after
a heart attack, you reprogram your heart cells to make
new cells like you did when you were in utero.
That is what we are exploring."
While the experiment shows promise, the
researchers acknowledge that their findings still won't be able to
prevent a heart attack.
However, they do hope to continue following up on
their findings to create therapies that can promote better mitosis
in heart cells. 8
Heart Muscles
Turn on Renewal Switches Under the Right Conditions
A related study published in Circulation also looked at how your
heart can make new muscle cells. The study tracked DNA signatures
inside cardiomyocytes to measure actual new cell formation, not just
cell enlargement. 9
According to the researchers, the goal was to determine whether the
adult human heart has what they called a "latent cardiomyocyte
regenerative potential" and whether certain conditions activate it.
The findings?
Your heart's ability to replace lost cells varies
dramatically depending on your physiological state, with some
patients showing dramatic surges in renewal when conditions improve.
-
Framework of the analysis
A group of patients with advanced heart
failure provided the data for this analysis.
The study
compared their heart tissue with healthy adult hearts and
then separated those who received left ventricular assist
device (LVAD) support - a mechanical pump that takes
workload off the heart - to see how different environments
affected cardiomyocyte renewal.
-
The enormous contrast between
healthy and failing hearts
In a normal adult, cardiomyocyte turnover
sits at about 0.5% per year, meaning a small but steady
replacement of muscle cells.
This results in an almost 40%
replacement during the entire lifespan of a human - a
contrast to the theory proposed in the earlier featured
study, wherein the number of cardiomyocytes remains the
same.
In end-stage heart failure, that renewal rate collapses. The
study reports that cardiomyocyte generation drops 18 to 50
times lower compared with healthy controls.
This means once
heart failure advances, your heart's natural repair
machinery slows to a crawl, making recovery harder unless
something shifts the internal environment dramatically.
-
A deeper look at the data
In failing hearts, renewal fell to 0.03%
per year for nonischemic cardiomyopathy and even to 0.01%
per year in ischemic cardiomyopathy - the type tied to heart
attacks.
This corresponds to the lower rate of regeneration
mentioned earlier.
Everything changes, however, in patients whose hearts
recovered function with LVAD support. Among those
individuals, cardiomyocyte renewal rose dramatically to 3.1%
per year.
This means some hearts aren't only stabilizing
under better conditions - they are rebuilding themselves at
a faster rate than healthy hearts normally do.
-
What's happening inside heart
muscle cells
The researchers documented that in the
worst heart failure cases, DNA synthesis inside
cardiomyocytes mostly produced polyploidy - extra DNA copies
inside the same cell - rather than creating entirely new
muscle cells.
In other words, your heart might look active at the
molecular level even while failing, but the activity is
misdirected.
Instead of replacing lost cells, the damaged
heart tends to enlarge existing cells or add extra nuclei, a
process that does not restore lost pumping strength.
-
A roadblock to regeneration
The researchers mentioned
cytokinesis
(the final step in cell division where one cell splits into
two) as a key chokepoint.
This means that many heart cells
are already entering the repair cycle, but they fail to
complete it. They copy DNA, they prepare to divide, but they
do not finish the split.
Your ability to rebuild heart
muscle depends on helping cells complete that final step.
-
Suggestions for future studies
While the researchers were able to detect
the regenerative rate in cardiomyocytes, they didn't go deep
into solutions.
However, they did offer suggestions that can
be used as a launching pad for other experts and expand
known facts in this field: 10
"[M]echanical unloading might reverse
metabolic cascades that increase reactive oxygen species
production.
This, in turn, can reduce oxidative DNA
damage and activation of the DNA damage response pathway
that causes cell cycle arrest in cardiomyocytes.
Indeed... a successful approach for cell replacement
strategies could be to selectively stimulate cytokinesis
in already cycling cardiomyocytes."
References
1 Centers
for Disease Control and Prevention
2 Mayo
Clinic, Heart Attack
3, 6, 7 The
Age, January 18, 2026
4 Circ
Res. 2026 Jan 16;138(2):e327486
5 Indica
News, January 20, 2026
8 Medical
Xpress, January 19, 2026
9, 10 Circulation.
2025 Jan 21;151(3):245-256
|