|
Far and away the most common question I get from those who took one of the COVID-19 vaccines is:
The mRNA and adenoviral DNA products were rolled out with no idea on how or when the body would ever breakdown the genetic code.
The synthetic mRNA carried on lipid nanoparticles appears to be resistant to breakdown by human ribonucleases by design so the product would be long-lasting and produce the protein product of interest for a considerable time period.
This would be an advantage for a normal human protein being replaced in a rare genetic deficiency state (e.g. alpha galactosidase in Fabry's disease).
However, it is a big
problem when the protein is the pathogenic
SARS-CoV-2 Spike. The
adenoviral DNA (Janssen) should broken down by deoxyribonuclease,
however this has not be exhaustively studied.
With the respiratory infection, Spike is processed and activated by cellular proteases including,
With vaccination, these systems may be avoided by systemic administration and production of Spike protein within cells.
As a result, the
pathogenesis of vaccine injury syndromes is believed to be driven by
accumulation of Spike protein in cells, tissues, and organs.
Out of all the available
therapies I have used in my practice and among all the proposed
detoxification agents, I believe nattokinase and related peptides
hold the greatest promise for patients at this time.
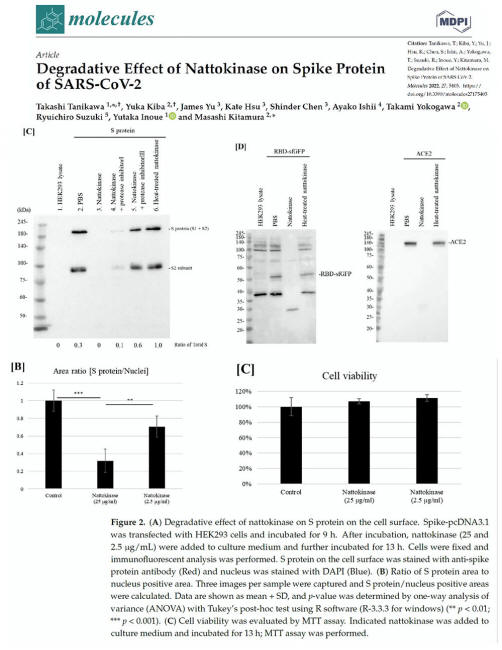
Tanikawa T, Kiba Y, Yu J, Hsu K, Chen S, Ishii A, Yokogawa T, Suzuki R, Inoue Y, Kitamura M. Degradative Effect of Nattokinase on Spike Protein of SARS-CoV-2. Molecules. 2022 Aug 24;27(17):5405. doi: 10.3390/molecules27175405. PMID: 36080170; PMCID: PMC9458005. Source:
Degradative Effect of
Nattokinase on Spike Protein of SARS-CoV-2
Kurosawa and colleagues have shown in humans that after a single oral dose of 2000 FU D-dimer concentrations at 6, and 8 hours, and blood fibrin/fibrinogen degradation products at 4 hours after administration elevated significantly (p < 0.05, respectively).
Thus an empiric starting dose could be 2000 FU twice a day.
Full pharmacokinetic and
pharmacodynamic studies have not been completed, but several years
of market use as an over-the-counter supplement suggests nattokinase
is safe with the main caveat being excessive bleeding and cautions
with concurrent antiplatelet and anticoagulant drugs.
The issue at hand is the urgency of time, similar to that with SARS-CoV-2 infection and empiric early therapy.
It will take up to 20 years to have a fully developed pharmaceutical profile to characterize the safety and efficacy of nattokinase in the treatment of vaccine injury and post-COVID syndromes.
Large number of people are sick now and many believe empiric treatment is justified given sufficiently low risk of side effects and potentially high reward.
My recommendation is to
discuss this with your doctor or seek a specialist in holistic or
naturopathic medicine who is experienced with the safety profile of
nattokinase in a range of applications.
|